Challenges of Conventional Drug Products
Dosing frequency
Maintaining patient adherence to dosing schedules, especially when a medication requires multiple doses per day, is a longstanding problem. A 2013 survey of patients with chronic conditions found that just 24% had perfect adherence to their dosing schedule, and the remaining 76% of patients either accidentally or intentionally failed to take their medication as directed. Of those who showed noncompliance, 57% said they missed a dose, and 30% said they couldn’t remember whether or not they took the dose at its scheduled time.

Figure 1: Statistic on patient compliance and medication dosages
Medication noncompliance is a particular problem among elderly patients, who are more likely to have a greater number of drugs to manage. Half of elderly patients who take only one medication a day find it difficult to adhere to the proper dosage schedule, and that proportion increases with the number of medications.
Tablet size
In addition to dosing frequency, the physical aspects of the drug product itself can have a dramatic impact on patient compliance. Tablet size is one example. A 2013 study by the European Journal of Clinical Pharmacology found that more than 37% of adults had difficulty swallowing pills, and 10% said they stopped taking medication as a result.
Large tablets present a barrier to use for any patient, but especially for diabetics, the elderly, and children. Elderly people naturally lose muscle mass and strength with age, which can impact their ability to swallow and increase the likelihood of choking. That risk carries over to medications, where large pills or capsules pose not only a deterrent to the geriatric patients taking them, but a choking hazard as well.
A similar problem can be found at the other end of the age spectrum. One pediatric study found that as many as 40% of children have difficulty swallowing a normal-sized pill.
Liquid-specific challenges
While tablets and pills may be a more common form of medication, liquid formulations pose their own set of challenges when developing a drug with patients in mind. For example, these formulations are most often administered by pouring the medication onto a spoon or into a small cup, but solutions that spill easily can make that difficult or lead to improper dosage. Thicker, more viscous medications are sometimes a solution and may spill less easily but can be unpleasant to swallow.
Liquid suspensions pose their own unique challenge – inconsistent drug dispersion and the need to “shake well before use”. Suspensions are prone to separation, as suspended particles either float to the top of the surrounding liquid or sink to the bottom. This creates an extra step to administration, as patients must shake the medication to redistribute the particles and ensure uniform drug dosing.
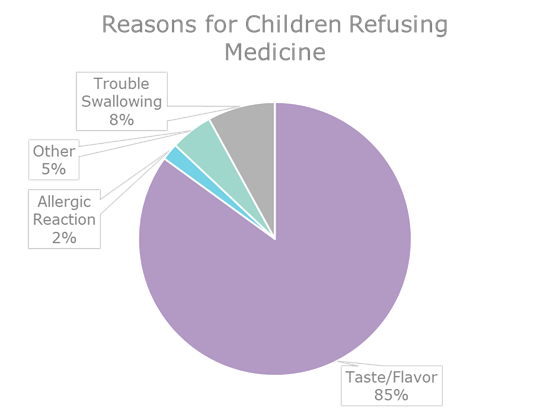
Figure 2: Pie chart on reported reasons children refuse medicine
Taste presents another challenge to liquid drug formulation, in particular for pediatric medications. Liquid drugs can be easier to administer to children, but bitter-tasting medications can make it difficult to ensure that children take them consistently and at the appropriate dose. Noncompliance can have serious and long-lasting impacts on patient health. In approximately half of all cases in which a drug has failed to have its desired effect in long-term treatment, patient noncompliance is a factor.
Patient-Centric Solutions to These Challenges
Though these challenges might feel daunting, there are numerous options drug developers can implore to overcome them, which we discuss below. Often the right excipient provides an easy solution, especially if partnering with a knowledgeable organization. A key step toward creating patient-centric medications is choosing an excipient specifically designed to impart critical consumer-driven properties to encourage compliance.
Extended Drug Release
Consistently, and across studies of various health conditions, medication regimens that require only a single daily dose see the highest compliance among patients. One study that prescribed antibiotics for respiratory tract infections found that those required to take a once-a-day medication dose were approximately 10 times as likely to comply as patients whose medication required two doses a day.
The most common solution is formulating drugs to have extended release properties. Extended release formulations have many advantages compared with immediate release formulations, including a reduction in the number of pills needed and the dosing frequency, as well as minimization of side effects caused by fluctuations in drug plasma levels.
For drug developers, the key to creating effective extended-release drug products lies largely in the excipients chosen during formulation. With the right excipient, tablets can be designed to better facilitate long release times, resulting in the same efficacy with fewer doses.
Carbopol® polymers are well known for their extended drug release capabilities because they are crosslinked polymers. Carbopol, being crosslinked, uses chemical bonds to entangle the drug, so it can provide longer controlled drug release at much lower concentrations than other polymers. Most other polymers are linear, like cellulosics, which means the drug is held in the polymer matrix only by weak physical entanglements. This results in a higher polymer concentration being needed and potentially more frequent dosing, making Carbopol a preferred choice for extended release formulations.

Figure 3: Quote on medication adherence
Pill Size Reduction
Excipients such as Carbopol present solutions to other formulation obstacles as well. Many oral tablets on the market today come in large sizes and can be hard to swallow. These pills are usually designed with linear polymers that require a greater amount of excipient to bind with the drug, resulting in a large tablet size. Carbopol, however, creates stronger bonds and requires less excipient, resulting in tablet size reduction and more patient-friendly products.
In one comparison of a commercial metformin tablet and a metformin tablet made using Carbopol at the same drug dose, the tablet containing Carbopol was over 20% smaller than its commercial counterpart. That level of size reduction can be significant for a patient with difficulty swallowing and can mean the difference between taking a medication properly or not.
No-Spill Rheology, Permanent Suspension & Taste Masking
Solutions formulated using Carbopol have shear thinning properties that help solve the problem of easy-to-spill liquids. Formulations containing Carbopol can be viscous at rest but become less so as they are poured or swallowed, before immediately becoming viscous again once pouring is complete. This means a patient will reliably be able to administer the drug product without fear of spillage.
Carbopol polymers’ properties also make them beneficial for stabilizing oral suspensions. Carbopol’s high yield value — its resistance to initial flow — means that it can create permanent, well-dispersed suspensions that do not separate, without requiring high viscosity which may be unpalatable to patients.
Carbopol polymers additionally solve the problem of unpleasant medication taste. Carbopol effectively masks the bitterness of a medication, without compromising the bioavailability of the drug as other methods can.
Conclusion
When a patient struggles to take a medication properly, the health and lifestyle consequences are important. Missed doses or wasted medication mean less-effective treatments and worse outcomes for end users.
When formulating drugs, patient safety and use should be top of mind for developers, as it allows them to differentiate from competitors. As technological progress induces patients to demand comfort and ease of use from their healthcare providers, they will increasingly look for the same in their medications.
Lubrizol maintains its position as a leader in creating safe, patient-centric excipients like Carbopol polymers, to the benefit of drug formulators and end users alike. For more than 50 years, Carbopol polymers have been trusted for their quality and safety. Contact our technical team for more information on how Carbopol polymers can help overcome your drug formulation challenges.